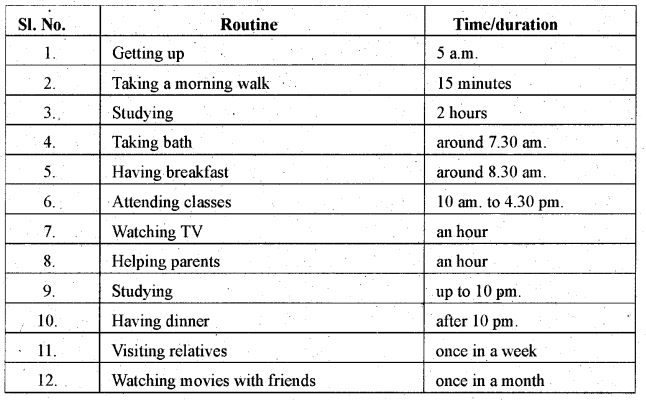
Karnataka 1st PUC English Workbook Answers Articulation Word Order
To make meaningful sentences words need to be arranged in a particular order.
A. I. Arrange the words to make affirmative sentences.
1. like /I/ you
2. French /I/ speak
3. hates / pigeons / he
4. The girls/song/a/sing
5. sell/flowers/we
6. you/to see/me/he/wants
7. feed/you/my/cat/can
8. sister/has/my/got/a dog
9. must/the book/read/you
10. buy/milk/he/wants/to
Answer:
1. I like you.
2. I speak French.
3. He hates pigeons.
4. The girls sing a song.
5. We sell flowers.
6. He Wants me to see you. / He wants you to see me.
7. You can feed my cat.
8. My sister has got a dog.
9. You must read the book.
10 . He wants to buy milk.
![]()
II. Arrange the words to make negative sentences. Place time expressions at the end of the sentences.
1. Our holiday / will / at home / we / not / spend / next year
2. did /I/ him / see / not / last night / at the disco
3. to a party / not / we / tonight / going / are
4. will / the letter / not / next week / send / you / she
5. not / the truth / did / he / tell / you / yesterday
6. to the cinema / we / want / not / do / tonight / to go
7. play / did / he / on Sunday / not / the piano
8. not / now/ she / inEngland / is
9. eat / in winter / ice cream / do / not /I
10. right now / have / not / we / time / do
Answer:
1. We will not spend our holiday at home next year.
2. I did not see him at the disco last night.
3. We are not going to a party tonight.
4. She will not send you the letter next week.
5. He did not tell you the truth yesterday.
6. We do not want to go to the cinema tonight.
7. He did not play the piano on Sunday.
8. She is not in England now.
9. I do not eat ice-cream in winter.
10. We do not have time right now.
![]()
III. Rearrange the segments to form questions.
1. accept the judgement / did the man / the jungle lords / of?
2. Mara tie / the creeper / to a tree / why did?
3. was the thief / his attitude / really humane / in / ?
4. does technology / in contrast / to religion / stand?
5. treated as / a / was Mara / mad man?
6. retain / did Douglass / his / self confidence?
7. why did / the farmer / suicide / commit?
8. the narrator / how does / go / to Verona?
9. what did / do / during the German invasion / the boys?
10. the crude realities of life / is love / of any value / before?
11. who / the window / broke? .
12. How/your grandmother/is ?
13 . did / she ./ her homework / in my room / do ?
14. Talaguppa Station / does / leave / the train / when?
15. conduct / classes / where / Babar Ali / does ?
16. has been cursed / which / a sage / creeper / by ?
Answer:
1. Did the man accept the judgement of the jungle lords?
2. Why did Mara tie the creeper to a tree?
3. Was the thief really humane in his attitude?
4. Does technology stand in contrast to religion?
5. Was Mara treated as a mad man?
6. Did Douglas retain his self confidence?
7. Why did the former commit suicide?
8. How does the narrator go to Verona?
9. What did the boys do during the German invasion?
10. Is love of any value before the crude realities of life?
11. Who broke the window?
12. How is your grandmother?
13. Did she do her homework in my room?
14. When does the train leave Talaguppa station?
15. Where does Babar Ali conduct classes?
16. Which creeper has been cursed by a sage?
![]()
B. 1. Rewrite the following sentences using the word / words in brackets.
1. No one knows about this. It’s a secret, (really)
2. She told him once, (only)
3. I eat a lot. (always)
4. Are you going to try? (hard)
5. He called you this morning, (many times)
6. I have been to Mumbai. I met my friend, (there)
7. I am not going to tell this story, (again)
8. Have you been to Chennai? (ever)
9. I have never tried this method, (before)
10. We saw a film, (last Wednesday)
11. The cat is playing? (in the garden)
12. They can remember, (never)
13. The old man waited for us. (eagerly)
14. Shake before use. (well)
15. We do not understand, (still)
Answer:
1. No one really knows about this. If a secret.
2. She told him only, office / She told him, only once.
3‘. I always eat a lot.
4. Are you going to try hard?
5. He called you many times this morning.
6. I have been to Mumbai. There I met my friend.
7. I am not going to tell this story again.
8. Have you ever been to Chennai?
9. I have never tried this method before.
10. We1 saw a film last Wednesday.
11. The cat is playing in the garden.
12. They can never remember.
13. The old man eagerly waited for us.
14. Shake well before use.
15. Still we do not understand.
![]()
Rearrange the juinbled segments to form meaningful sentences, we / some interesting books / found / in the library
Shyam / play football / does / every weekend
are building / opposite the park / a new: hotel / they
his name III remembered / after a few minutes
please write your name / at / beginning / of / page / the / the
go to / I / every Friday / the mosque
so late / did / come home / why / you
London / lam going / to / next week / for / a few days
on / at / Saturday evening / you / didn’t see /1 / the temple
around / the procession / the Church / went
Answer:
1. We found some interesting books in the library
2. Does Shyam play football every weekend?
3. They are building a new hotel opposite the park.
4. I remembered his name after a few minutes.
5. Please write your name at the beginning of the page.
6. I go to the mosque every Friday.
7. Why did you come home so late?
8. I am going to London for a few days next week.
9. I didn’t see you at the temple on Saturday evening.
10. The procession went around the Church.